Antarcticite Structure
Steven Dutch, Professor Emeritus, Natural and Applied Sciences, Universityof Wisconsin - Green Bay
Antarcticite, CaCl2.6(H2O) is one of the "juiciest" musherals, having twice as many water molecules as other atoms. I recall calcium chloride from my chemistry set as a kid. It had a nasty medicinal smell and a tendency to harden into a solid useless mass by hydration. Calcium chloride is a common ice melter, since it releases heat when it dissolves and works at lower temperatures than ordinary salt.
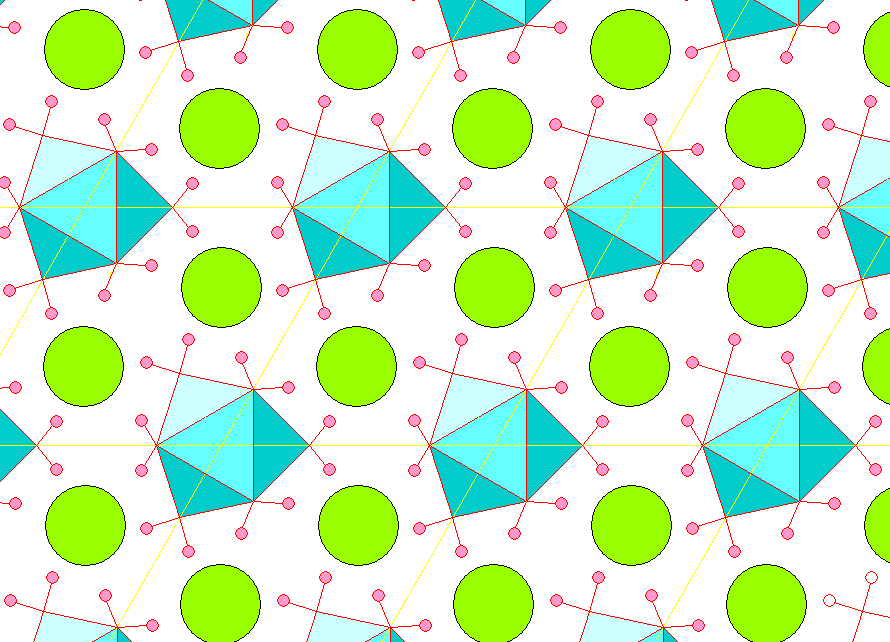
The structure is easy to visualize and really pretty if you like symmetry and polyhedra. It consists of Ca atoms surrounded by nine water molecules, with the complex cations linked by Cl atoms. A view of the structure is shown below, with the Mg-H2O coordination polyhedra shown in blue. Hydrogen is pink and chlorine is green. Unit cell edges are yellow.
Below is a depiction of the atoms. Oxygen atoms are blue (those in the lower layer are darker). Calcium atoms are purple, peeping through interstices between the upper water molecules, hydrogen is pink and chlorine is green. In reality, the hydrogen atoms should be much bigger.

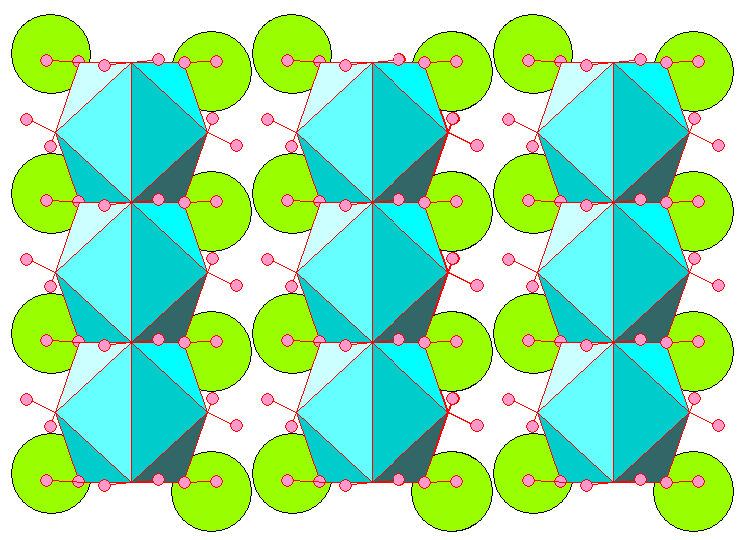
Below is a view with the c-axis (threefold symmetry) vertical, looking at 30 degrees to the unit cell edges, showing the Ca-H2O coordination polyhedra.
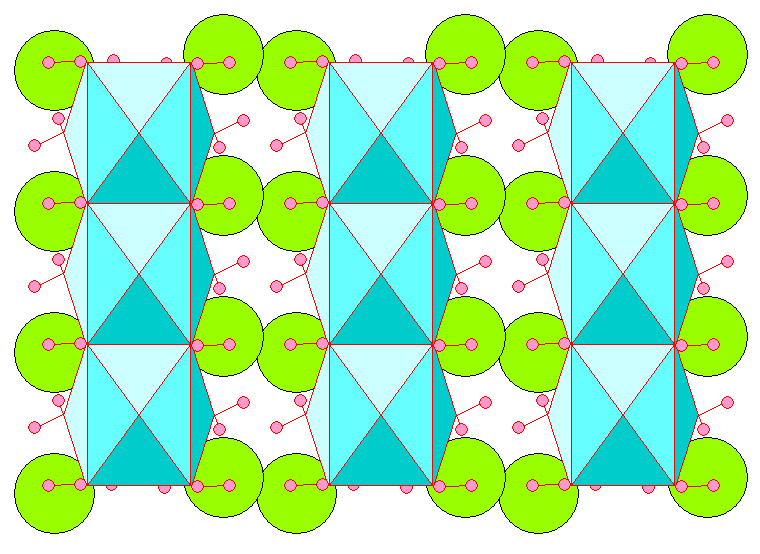
Below is a view with the c-axis (threefold symmetry) vertical, looking along the unit cell edges, showing the Ca-H2O coordination polyhedra.
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 22 Sept 1997, Last Update 16 December, 1999