Apophyllite Structure
Steven Dutch, Professor Emeritus, Natural and Applied Sciences,
Universityof Wisconsin - Green Bay
Apophyllite is a phyllosilicate, but the silica sheets are thicker and more three-dimensional than other phyllosilicates. It does not crystallize as platy crystals and occurs with zeolites rather than other phyllosilicates. Thus, it tends to be placed at the tail end of the phyllosilicate sections of references, just before the framework silicates. Actually, because of the variations in cation content and replacement of hydroxyl by fluorine, apophyllite is now used as a mineral family name rather than a single mineral.

The view above shows the silica sheets, consisting of four-membered rings alternately pointing up and down and enclosing flattened octagonal rings.
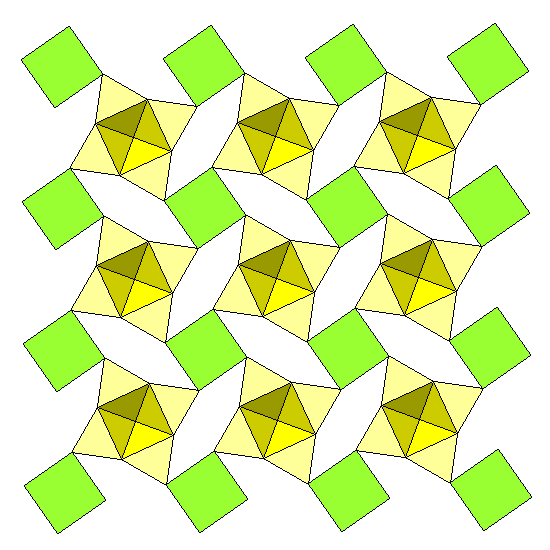
The cation sheets consist of cubes of potassium atoms enclosed by water molecules, and calcium polyhedra. The calcium polyhedra are "augmented triangular prisms," consisting of a triangular prism with a square pyramid built out on one face. The outer corners of the polyhedron are water molecules, the atoms around the base of the pyramid are oxygens shared with silica tetrahedra, and the apical ion at the center of the square dimple is a hydroxyl (OH). Four calcium polyhedra surround the central dimple.
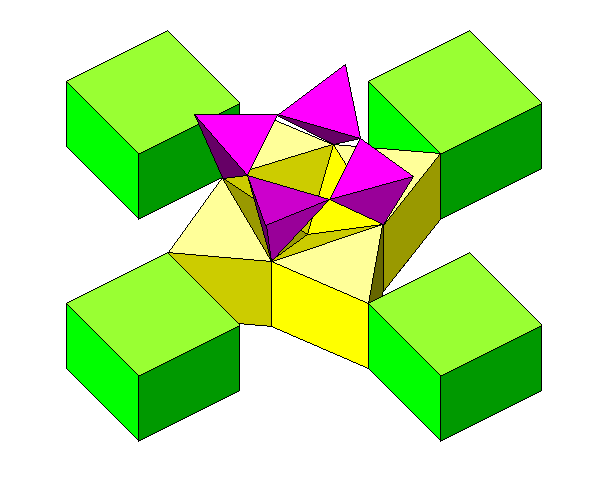
This view shows how silica tetrahedra mate with the calcium polyhedra

The view above shows a silica sheet overlying a cation sheet.

The view above shows hydrogen atoms in red and the hydroxyl ions in blue. H-O bonds in water molecules are shown in dark green (one hydrogen is superimposed above its corresponding oxygen atom) and hydrogen bonds to other oxygen atoms are in light green.

Above is an edge-on view of the apophyllite structure. Potassium polyhedra are in green and calcium polyhedra in yellow.
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 22 Sept 1997, Last Update 22 Sept 1997