Bischofite
Steven Dutch, Professor Emeritus, Natural and Applied Sciences, Universityof Wisconsin - Green Bay
Bischofite MgCl2*6(H2O), Nickelbischofite NiCl2*6(H2O) and Antarcticite CaCl2*6(H2O)have very similar formulas. All consist of a divalent cation electrostatically balanced by two chlorine atoms, plus six water molecules, or two water molecules for every other atom. Nonetheless they have quite different structures. Bischofite and Nickelbischofite both have cations octahedrally coordinated, but Antarcticite has its cation in nine-fold coordination with water molecules, probably due to the much larger ionic radius of calcium.
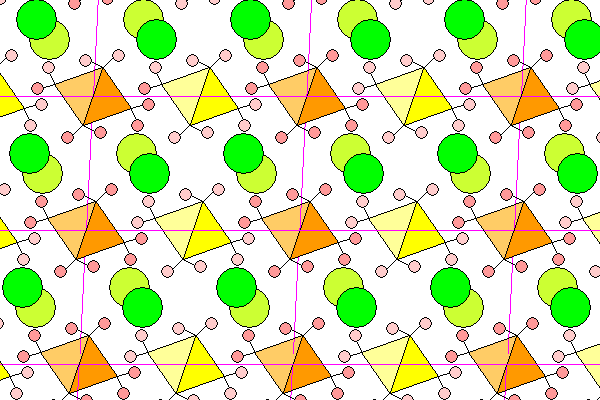
Bischofite misses being orthorhombic by the slimmest of margins. The a and c axes miss being perpendicular by less than four degrees. In this view, looking down the b axis (2-fold symmetry axis), the unit cell edges are magenta, the octahedral magnesium cage is yellow or orange, hydrogen atoms are pink and chlorine is green. At the center of each octahedron is a magnesium atom and each vertex is occupied by the oxygen atom of a water molecule. Foreground objects are in darker colors.
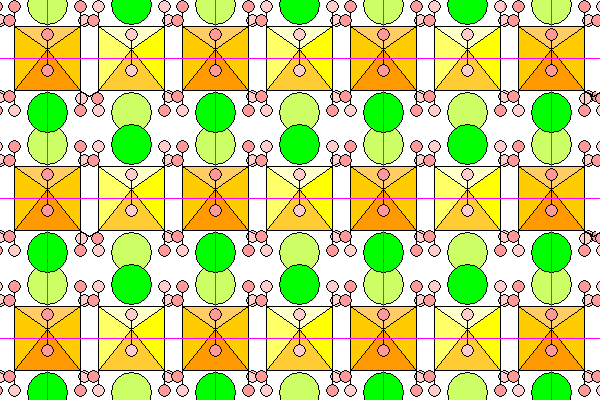
This view down the a axis shows the octahedra end on.
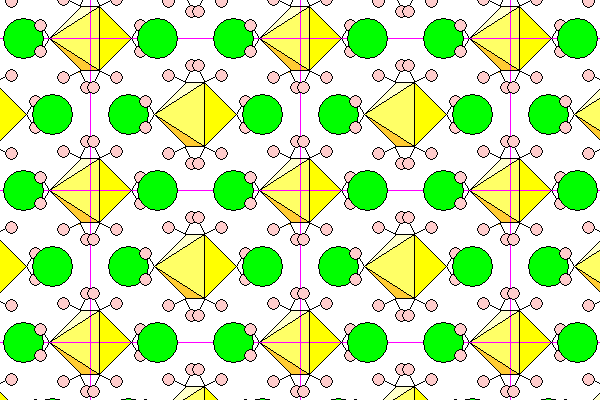
A view down the c-axis. Only one color tone is used since the octahedra are in the same plane and the chlorine atoms can be distinguished by their overlap with the hydrogens.
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 21 March 2011, last update