Geochemical Associations
Steven Dutch, Professor Emeritus, Natural and Applied Sciences, University of Wisconsin - Green Bay
We can recognize a number of groups of elements that tend to occur together in rocks and minerals, because of chemical similarities, ionic charge, or ionic radius.

- Lithophile ("rock loving") elements occupy the left side and right top of the periodic table and tend most often to occur as silicate minerals.
- Siderophile ("iron loving") elements occupy the left center of the periodic table. They tend to occur as oxides, are abundant in iron meteorites, and tend to be concentrated in the Earth's mantle and core.
- Chalcophile ("brass loving") elements occupy the right center of the periodic table. They tend to form sulfides and are are also enriched in the Earth's interior.
- Platinum Group elements are usually regarded as part of the siderophile group, but they are shown here separately because they are the elements most likely to occur in the metallic form in nature, usually as natural alloys rather than pure elements.
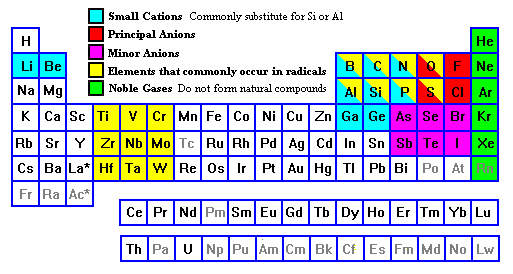
- Noble Gases have complete outer octet shells. They can be made to combine with other elements in the laboratory but have not been found in compounds in nature.
- Principal Anions include oxygen, far and away the most important anion in the Earth, followed by sulfur, with chlorine and fluorine trailing distantly.
- Minor Anions include the other two halogens, bromine and iodine, plus some of the semi-metals. They are uncommon and the semi-metals can also occur as cations.
- Radical-formers include a cluster at the top right of the periodic table (easily the most important group) plus another cluster on the left side of the transition elements.
- Small Cations include Li+1, Be+2, B+3, C+4, N+5, Al+3, Si+4, P+5, Ga+3, and Ge+4, with ionic radii smaller than 0.5 Angstrom units. Their small sizes enable them to substitute for Si or Al, or cause them to form compounds with different packings than compounds of elements further down the columns. The wide range of ionic charges means they don't always substitute for one another: Li can sometimes substitute for Al but rarely for Si, for example.
- Large Cations include Na, Mg, and the elements beneath them, plus a few others with +1 or +2 charge, notably Sn+2 and Eu+2. These are typically about 1 Angstrom Unit in diameter. Europium is a particularly interesting case. The rare earths are so similar that they tend to occur interchangeably in minerals, but because Eu is a close twin in size and charge to Ca and Sr, it tends to substitute for those elements. Thus, if Eu is conspicuously enriched or depleted compared to other rare earths, it signals major movements of Ca or Sr, such as removal or accumulation of feldspar in a magma. Note also that Li and Be are not part of this group. The charges and chemical affinities are right, but they are simply too small to occupy the sites that large cations do. Li would simply slip through the holes in a feldspar structure, for example.
- Large Cations of High Charge include the block with corners at Sc and W plus a few others like Pb+4.
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 18 September 1998, Last Update