Sodalite Structure
Steven Dutch, Professor Emeritus, Natural and Applied Sciences, University of Wisconsin - Green Bay
The Sodalite group of minerals consists of:
- Sodalite Na4(Al3Si3)O12Cl
- Nosean Na8(Al6Si6)O24(SO4).H2O
- Lazurite Na3Ca(Si3Al3)O12S
- Hauyne: solid solution of sodalite and nosean
If nepheline and leucite can be called feldspathoids, because they are chemically similar to the feldspars and occupy the same "ecological niche," we can perhaps call the sodalite group "scapolitoids." They are chemically similar to feldspathoids but contain anions other than SiO4.
All the sodalite minerals are isometric. The structure consists of a network of tetrahedra in each unit cell outlining a truncated octahedron. A ring of four tetrahedra surrounds each four-fold symmetry axis, and these link together to form six-sided rings around each three-fold axis. Additional anions sit in the center of each unit cell and alternate corners.
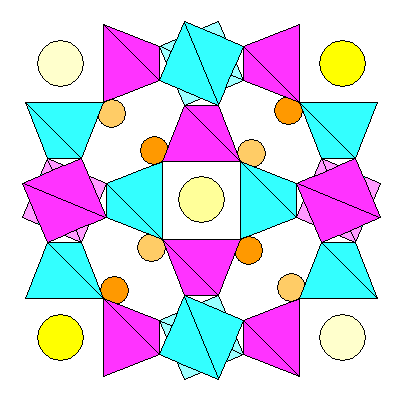
View of a sodalite unit cell down a fourfold axis. Silica tetrahedra are purple, AlO4 tetrahedra are blue, Sodium (or other cation) atoms are orange and Chlorine (or other anions) are yellow.
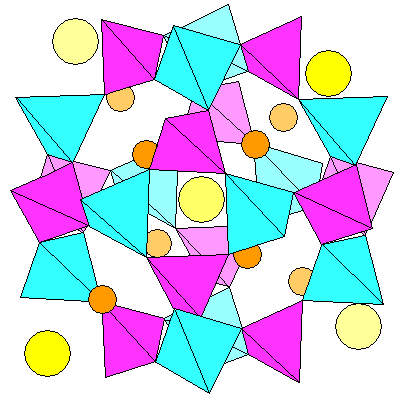
Slightly oblique view showing front and rear of the structure. In both views, lighter colors indicate atoms or tetrahedra in the background.
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 15 October 2009, Last Update