Staurolite Structure
Steven Dutch, Professor Emeritus, Natural and Applied Sciences, University of Wisconsin - Green Bay
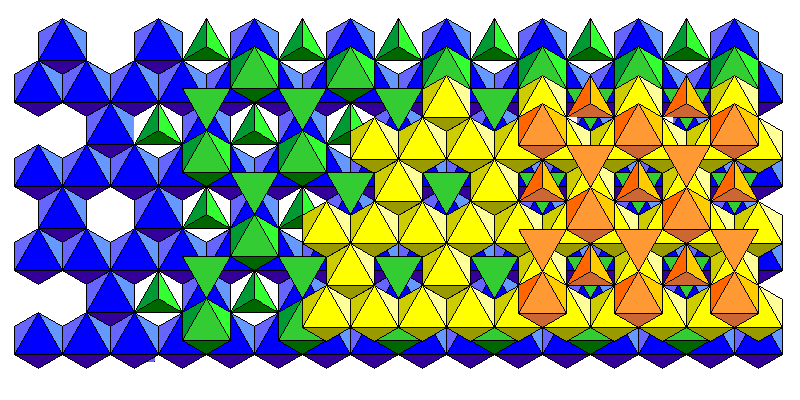
The staurolite structure consists of oxygen atoms in an approximately cubic close packing. However, the cation arrangement does do not have cubic symmetry. The structure consists of dioctahedral sheets joined by isolated octahedra. Silica tetrahedra fill the gaps between octahedra. A polyhedral model is below.
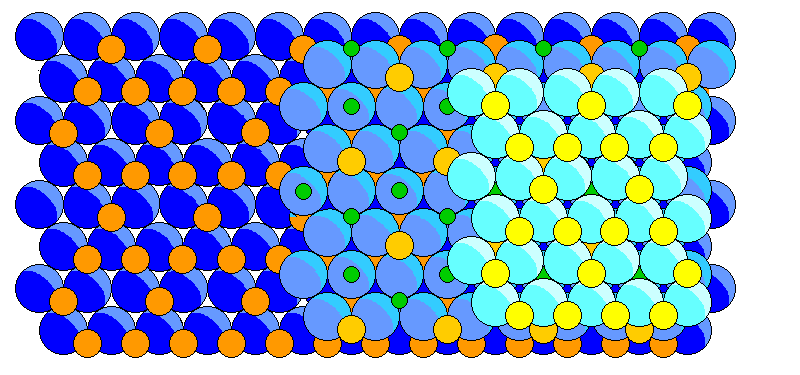
below is a ball model of staurolite. Blue atoms are oxygen, yellow and orange are iron or magnesium, and green are silicon.
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 15 October 2009, Last Update