Scale Drawings of Atoms and Orbitals: Hydrogen Through Krypton
Steven Dutch, Professor Emeritus, Natural and Applied Sciences, University of Wisconsin - Green Bay
Although almost any chemical reference will have data on ionic radii and electron configuration, and tabulations of energy levels are not hard to find, there is little readily available data on the relative sizes of orbitals. Ionic radii are useful in crystal modeling, electron configuration and energy levels for all kinds of stuff, but sizes of orbitals don't seem to have much practical use. Principally they come out of theories whose main purpose is to account successfully for the other properties of atoms. These drawings are based on data from J. T. Waber and D. T. Cromer, Orbital Radii of Atoms and Ions, Journal of Chemical Physics, 15 June, 1965, vol. 42, no. 12, p. 4116-4123.
All the drawings are to common scale. For orbitals with lobes, the principal lobes are shown but there is no attempt to portray the complex inner structure. It gets plenty busy in toward the center of the atom anyway. In any case, we really don't know in detail what the orbitals deep in an atom look like anyway. All the pictures in textbooks are for single electron atoms. Interactions with other electrons and the screening effect of electrons closer to the nucleus will affect the shapes of orbitals in complex ways.
Note that the orbitals are fuzzy, not sharp-edged! That means the presence of electrons extends beyond the orbitals shown. In fact well beyond, since the radii are the radius of maximum electron density. That distinction will become important when we look at ions.
Hydrogen through Neon
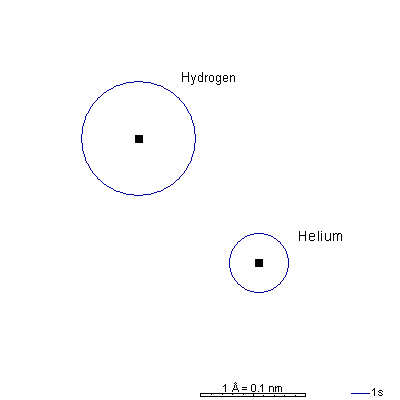 |
Hydrogen has a single electron in a 1s orbital.
This is the only atom for which Bohr's planetary model gave accurate results. The orbital radius, 0.53 Angstroms or 53 picometers, is sometimes called a "Bohr unit" and orbital dimensions are often tabulated in Bohr units rather than metric, especially in older literature. That unit system is often called "atomic units." Helium also just has a 1s orbital. It has two electrons but also two protons. So the stronger attraction of the nucleus makes the helium s orbital only a little more than half the radius of the hydrogen s orbital. |
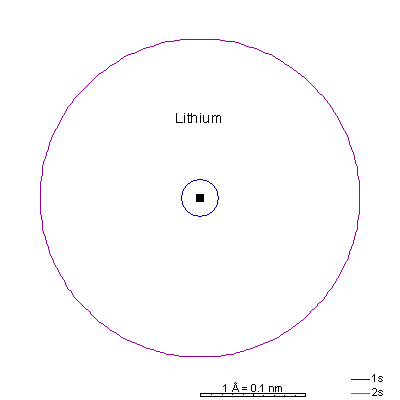 |
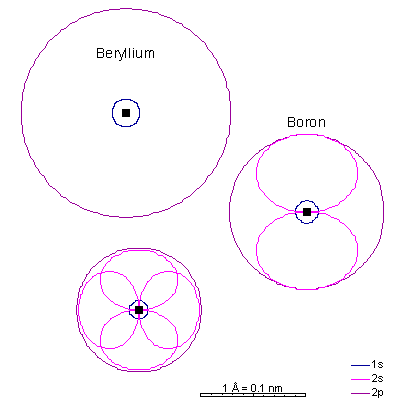
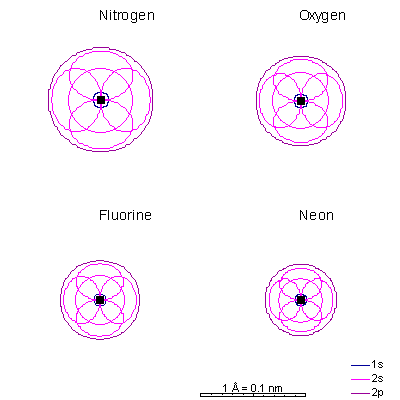
Lithium and beryllium have a 2s as well. The shielding effect of the 1s electrons is the same as in helium but there are three protons in lithium. The 1s radius is about a third that of hydrogen. Ionizing the elements lithium through carbon strips off the 2s and 2p electrons, leaving only the 1s shell and creating extremely small cations. Ionizing the elements nitrogen through fluorine fills the p orbitals, causing the ion to "puff up" because the electron repulsion can partially overcome the attraction of the nucleus. With only 7, 8, or 9 protons, an extra electron makes a big difference. The result is fat anions. Neon is the first element with a stable octet with complete p and s orbitals. The reason the octet is so stable is that the only way to add electrons is to add the next s orbital outward. There is no way to add electrons to the 2 shell of neon, period. |
 |
 |
period 3 (sodium to argon)
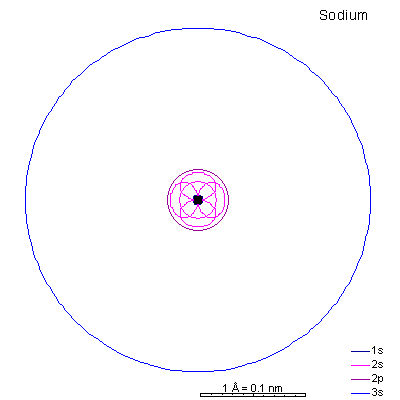 |
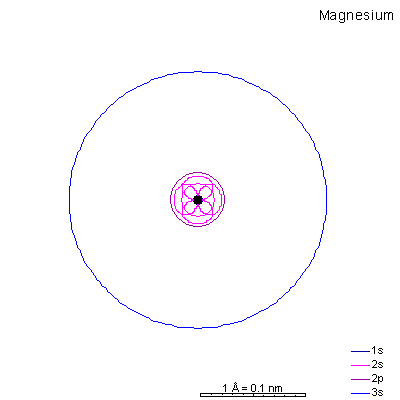
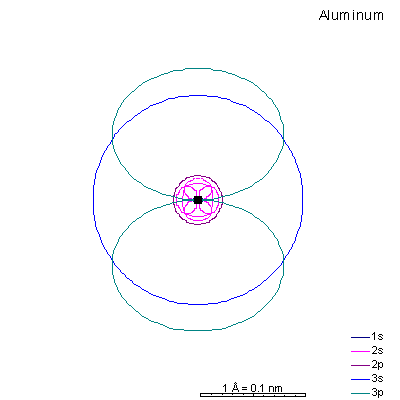
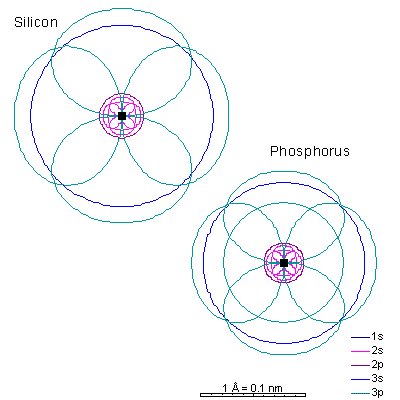
With sodium, we add a 3s orbital and at aluminum we begin adding 3p orbitals. Note that the 3p orbitals are larger than the 3s orbital.
This will be a general pattern: f orbitals are bigger than d orbitals, which are larger than p, with s as the smallest. With increasing atomic number and nuclear charge, the discrepancy diminishes. Orbitals in shells deep within the atom are pretty similar in size. As atomic number increases, the orbitals shrink dramatically in size as the positive charge in the nucleus increases. Argon is the next element with a stable octet with complete p and s orbitals. The reason the octet is so stable is that the only way to add electrons is to add the next s orbital outward. It is possible to add 3d orbitals, but not until the 4s orbital is full. So it's all but impossible to make reactive ions out of the noble gases. |
 |
 |
 |
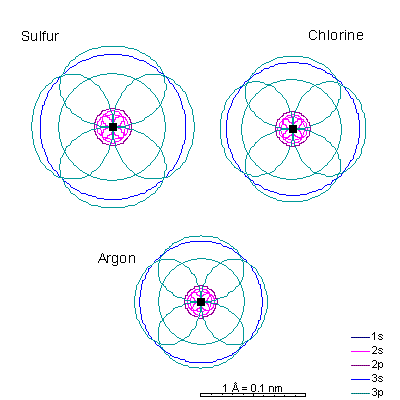 |
Period 4 (Potassium through Krypton)
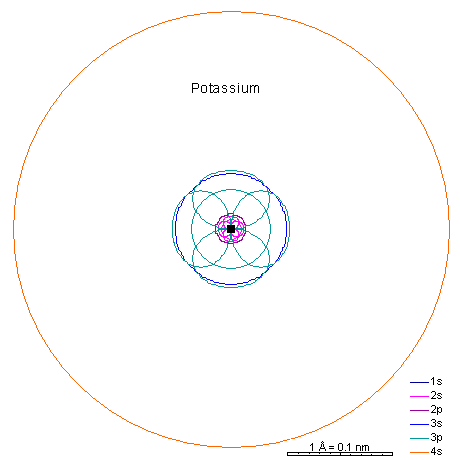
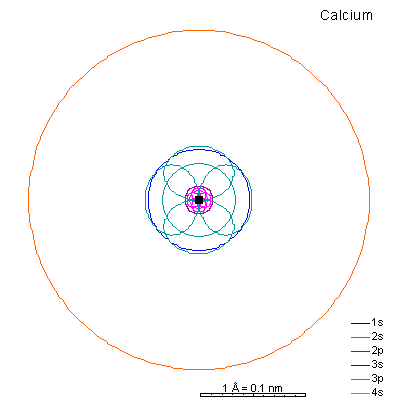 |
With potassium and calcium we begin adding the 4s shell. |
Transition Metals (Scandium through Zinc)
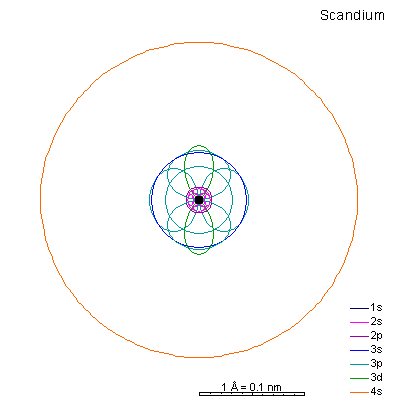 |
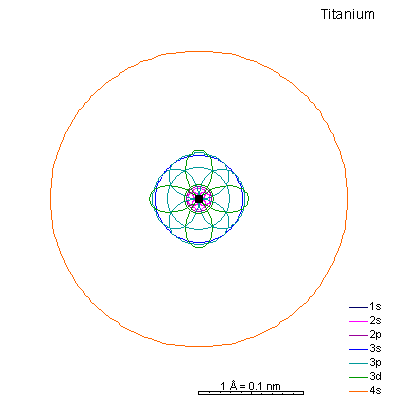
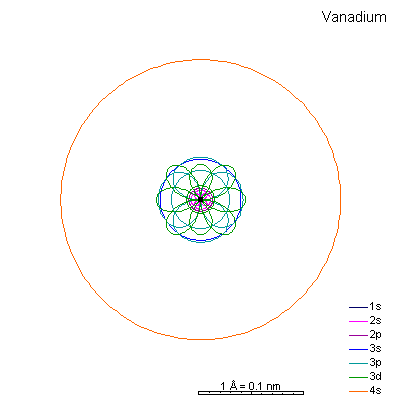
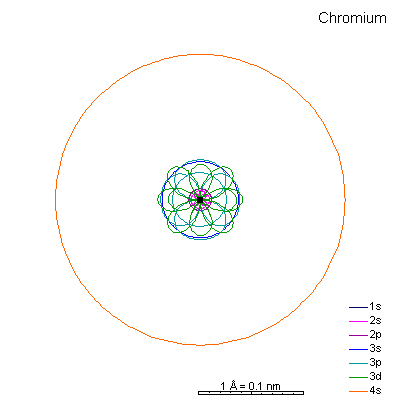
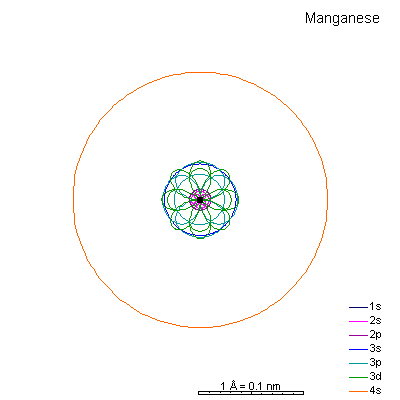
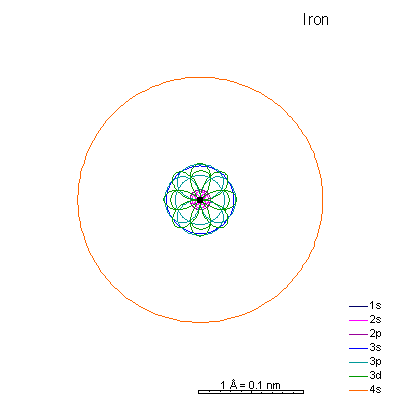
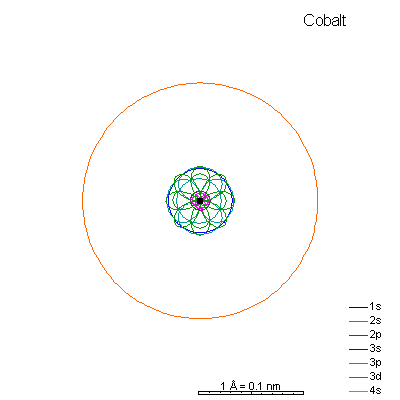
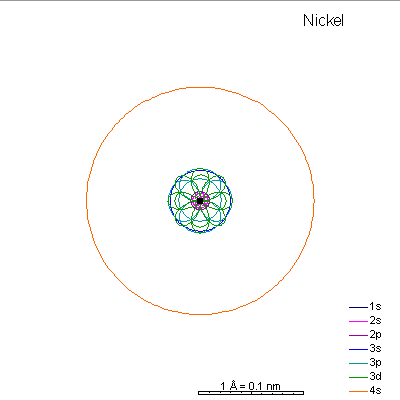
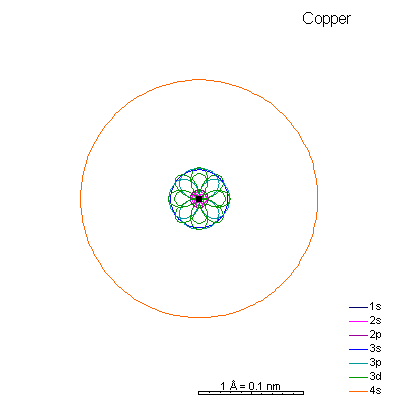
By now the 3p orbitals are almost the same radius as 3s. We begin adding 3d orbitals, which are noticeably larger. In this perspective, some of the 3d orbitals line up in the same planes as others and only very limited attempts will be made to try to show the overlaps.
We can see that ionizing these elements will often strip off the 4s shell, leaving rather small ions with +2 charge. Scandium through manganese can lose some or all the d electrons also, giving charges of +3 to +7, respectively. |
 |
 |
 |
 |
 |
 |
 |
 |
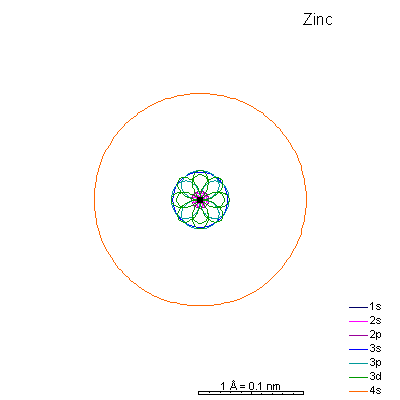 |
From iron on the most likely route for ionization is to lose the 4s electrons. We end up with medium-sized cations that generally have +2 charges. Given the charge and size similarities, it's hardly a surprise that there's a high (though not complete) degree of interchangeability of these ions in crystals. There is a systematic decrease in size with increasing atomic number, so Mn+2 is a lot more likely to substitute for Fe+2 than for Zn+2. |
Semi-Metals and Non-Metals (Gallium through Krypton)
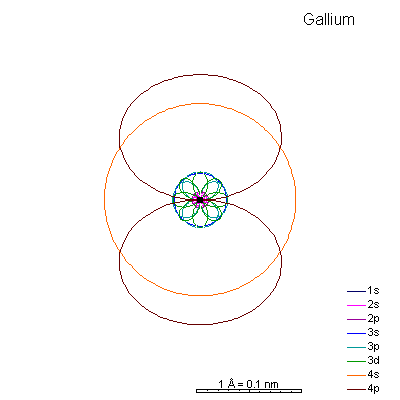 |
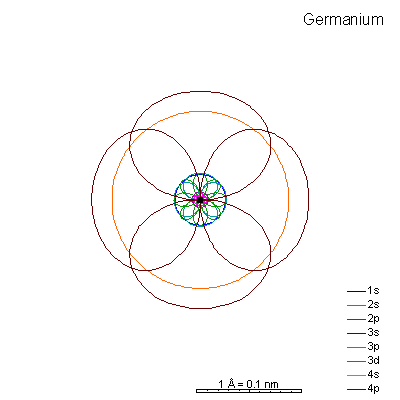
Shell 3 is completely full and we start adding 4p electrons. The 3p orbital radii are quite a bit bigger than the 4s.
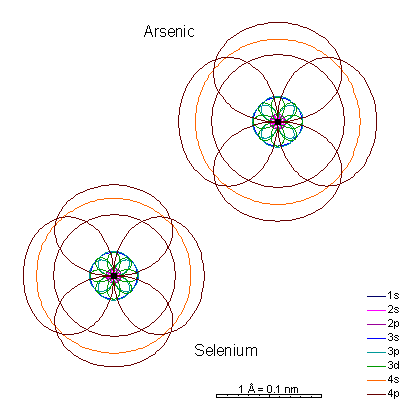
Ionizing gallium and germanium strips away the 4 shell electrons, creating very tiny cations. Arsenic and selenium can lose electrons to become cations, or gain electrons to become anions. |
 |
 |
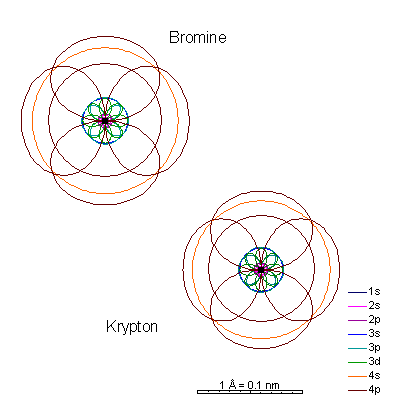 |
Bromine gains a last p electron to become an anion. |
What Do Atoms Really "Look Like?"
What Atoms of Hydrogen Through Xenon Really "Look Like"
What Atoms of the Heavy Elements Really "Look Like"
Scale Drawings of Atoms and Orbitals: Hydrogen Through Krypton
Scale Drawings of Atoms and Orbitals: Rubidium Through Xenon
Scale Drawings of Atoms and Orbitals: Cesium Through Radon
Scale Drawings of Atoms and Orbitals: Francium Through Lawrencium
What the Atomic Structures of Some Simple Materials Really "Look Like"
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 26 April 2006, Last Update